Uncovering the basis for microtubule-dependent plant stem cell maintenance
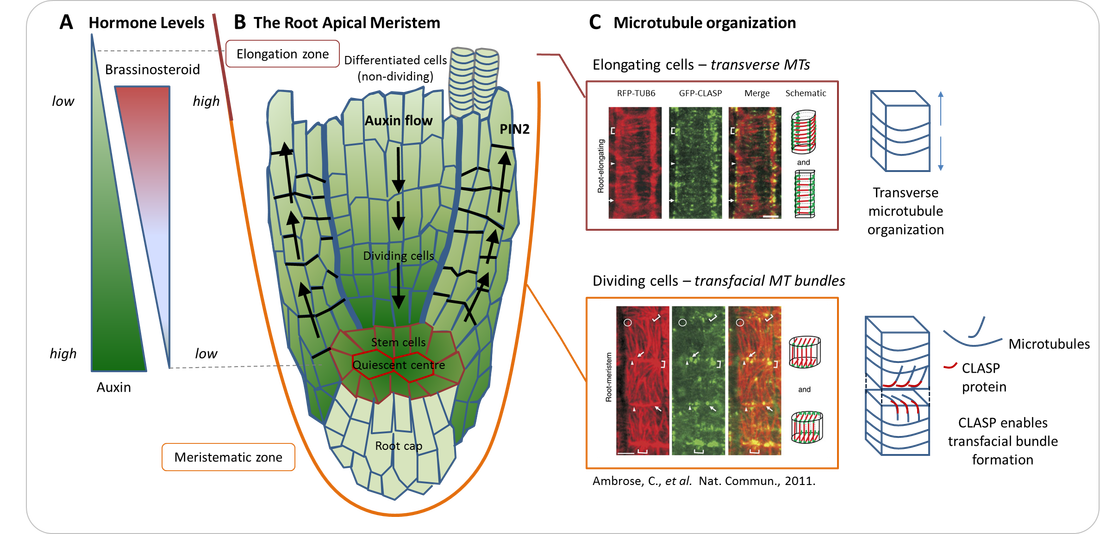
Growth in plants occurs at shoot and root apices, with new tissues arising from stem cell centres known as meristems, areas analogous to animal stem cell niches. A cartoon of the root apical meristem can be found in Figure A. Meristem size is tightly regulated and depends on a balance between the rate of uncommitted stem cell proliferation and cell differentiation into specific tissues. Various hormones play an important role in this regulation, but recent discoveries indicate that the cytoskeleton also plays an important part, with microtubules (MTs) regulating the hormone auxin and its transport via complex feedback mechanisms [1]. In addition, the organization of microtubules in dividing cells has been implicated in meristem maintenance, with the Arabidopsis thaliana MT-associated CLASP protein playing a critical role [2, 3].
CLASP accumulates at specific cell edges, enabling MT growth around these edges and promoting the formation of MT bundles that span adjacent cell faces [4]. Unlike the transverse conformation MTs typically adopt in elongating cells (Figure B), CLASP promotes transfacial MT bundle formation (Figure C). These transfacial bundles are strongly associated with maintaining the capacity for division, which is an important stem cell feature.
To characterize the developmental switches that lead to changes in MT organization a correlative imaging approach is necessary. High pressure freezing, freeze substitution and imaging with fluorescence light microscopy (fLM) followed by transmission electron microscopy (TEM) or tomography will enable the structural analysis of MTs at Arabidopsis thaliana cell edges at high resolution and in three dimensions.
CLASP accumulates at specific cell edges, enabling MT growth around these edges and promoting the formation of MT bundles that span adjacent cell faces [4]. Unlike the transverse conformation MTs typically adopt in elongating cells (Figure B), CLASP promotes transfacial MT bundle formation (Figure C). These transfacial bundles are strongly associated with maintaining the capacity for division, which is an important stem cell feature.
To characterize the developmental switches that lead to changes in MT organization a correlative imaging approach is necessary. High pressure freezing, freeze substitution and imaging with fluorescence light microscopy (fLM) followed by transmission electron microscopy (TEM) or tomography will enable the structural analysis of MTs at Arabidopsis thaliana cell edges at high resolution and in three dimensions.
Mathematical modelling is an invaluable tool for integrating information gained from different sources to make predictions about system functioning. Combining molecular data, live imaging dynamics, and my ultrastructural analysis, my plan is to create a virtual root apical meristem to characterize the role of the microtubules and CLASP in meristem maintenance. My model will integrate gene regulatory network modelling on the single-cell scale into a multicellular context.